Mostly in HILIC conditions, by increasing column temperature a decrease in the retention time is observed.
Column temperature is an important parameter in HILIC separations and affects the mobile phase viscosity, the solute diffusivity and the energy of the analyte transfer between mobile phase and stationary phase. Generally in HILIC, a decrease in the retention time is observed, increasing column temperature. As we have already discussed, HILIC retention mechanism is largely based on partitioning. The transfer of hydrophilic analytes from the hydrophobic bulk mobile phase at high acetonitrile content to the hydrophilic water layer is an exothermic process. Therefore, it is favoured at lower temperatures (Figure 1). The optimization of column temperature can improve analysis performance, executing faster runs at higher temperatures.
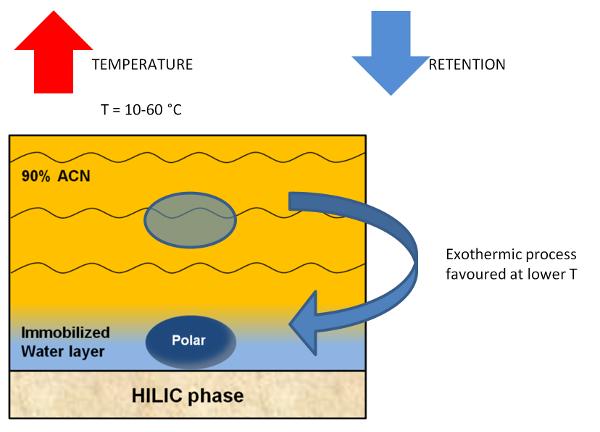 Figure 1. Schematic representation of the analyte transfer from the mobile phase to the HILIC stationary phase. In most cases this process is favoured at lower temperatures.
Figure 1. Schematic representation of the analyte transfer from the mobile phase to the HILIC stationary phase. In most cases this process is favoured at lower temperatures.
The main exception to this general behaviour is represented in the case of separation of basic analytes on silica columns or acid analytes on amino columns. In presence of strong electrostatic attractions between charged analytes and charged stationary phases, increasing the column temperature leads to an increase in the retention. The explanation of this phenomenon is based on the consideration that the formation of an ion pair in a aqueous solution between two oppositely charged groups is an endothermic process, supported athigher temperatures (Figure 2).
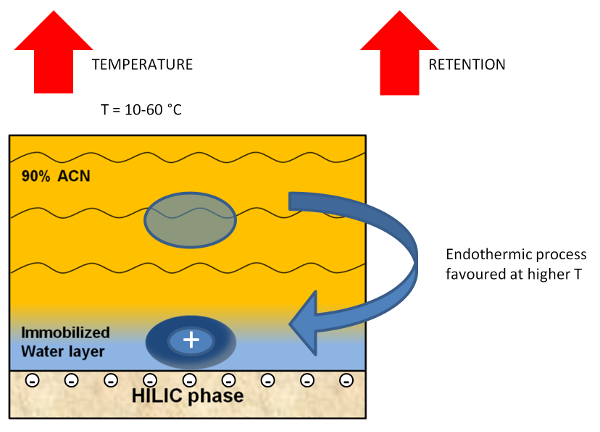
Figure 2. Schematic representation of a charged analyte transfer from the mobile phase to the HILIC stationary phase. The formation of an ion pair is favoured at higher temperatures.
Different types of compounds in a mixture may present different contributions to the retention mechanisms, more partitioning or more electrostatic interactions. Therefore, column temperature can affect also their selectivity, leading to a variation of the elution order.
The increase of column temperature can increment the analyte conversion rate between more configurations. For example, carbohydrates and similar compounds exist in an aqueous solution as a mixture of several tautomeric forms, leading to broad or separated peaks. The increase of column temperature has the effect of merge the different forms into single narrow peaks.
In HILIC, column temperature is usually set between 10 and 60°C. Higher temperature values can be used depending on both stationary phase stability and analyte stability.
It is worth to point out that the variation of temperature has a less impact on retention than the composition of the mobile phase. During the development of a HILIC method the chromatographic parameters should be optimised in the following order: (1) organic content in the mobile phase, (2) salt concentration and pH of the mobile phase, (3) column temperature.
In the next instalment, we will discuss the effect of column equilibration on HILIC retention and reproducibility.
This blog article series is produced in collaboration with Dr Giorgia Greco, Product Manager with Thermo Fisher Scientific in Germany and Thomas Letzel, Associate Professor and Head of the Analytical Research Group at the Technische Universität München, Germany.
Giorgia Greco received a PhD in Chemistry and worked as a Post Doc researcher at the Technische Universität München, Germany. During her research, she specialized in the fundamental of LC-MS and in the separation and analysis of metabolites from human and food matrices, as well as organic contaminants in waste water samples, by hyphenated HPLC/MS and HILIC/MS techniques.
Thomas Letzel received his PhD in Chemistry with Aerosol Analysis and then worked as a Post-Doc performing pharmaceutical analysis. He is the author of more than 50 publications and two books and wants to share his experience in liquid chromatography, especially in HILIC, with the community to accelerate the dissemination about HILIC theory and practical handling.
If you have any questions about this article send them to techtips@sepscience.com
